
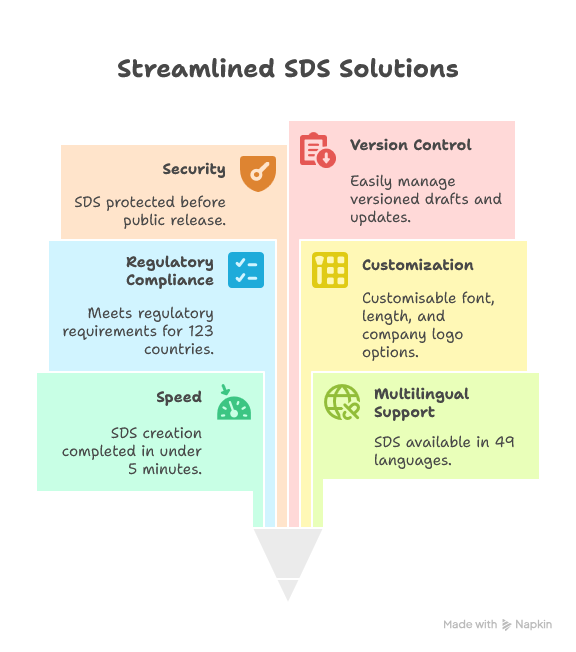
AuthorITe is the easy way to author SDS for the products you manufacture.
Packed with pre-classified substances, helpful suggestion tools and a step-by-step method for authoring, archiving and updating your SDS, AuthorITe meets the needs of the modern chemist.




Chemwatch has been a leading provider in chemical management systems for over 30 years. Specialising in chemicals safety, we're an international company headquartered in Australia, with offices throughout Europe, the US and Asia-Pacific regions. We are a large employer of science graduates and postgraduates—including chemists, toxicologists and OHS specialists. Thousands of organisations use Chemwatch services globally, including manufacturers, multinationals, hospitals, research institutes, and governments, for Chemicals Management, SDS Management and Authoring, and Regulatory Compliance.